You might have heard that your local water supply is hard – but what does that mean? What’s the difference between hard and soft water, and which is best?
We’ve answered this question in this hard Vs soft water guide.
Table of Contents
📌 Key Takeaways
- Hard water is water with a high mineral content.
- Soft water, on the other hand, contains very few or no minerals.
- Many folks soften their home’s water because hard water leaves damaging mineral deposits in pipes and appliances.
🆚 Hard Water Vs Soft Water Quick Comparison
Just looking to quickly compare hard and soft water by taste, properties, and more? This hard or soft water comparison should be useful.
| Hard Water | Soft Water | |
|---|---|---|
| Taste | Pleasantly alkaline and its minerals improve water’s taste. | May taste “plain” due to its lack of minerals, and salt-softened water may taste slightly salty and have a slippery texture. |
| Properties | Contains around 60-180+ GPG calcium & magnesium | Low magnesium and calcium content. |
| Health Effects | Healthier to ingest but causes dry hair and skin. | Doesn’t contain minerals, so it’s not as healthy as hard water, but it’s kinder to skin and hair |
| Home Effects | Damaging to a plumbing system and leaves scale deposits. | No major effects in the home, although it may have mildly corrosive effects in your water heater |
🚱 What is hard water?
Hard water is water that contains dissolved minerals like calcium and magnesium.
The higher the mineral content, the harder the water.
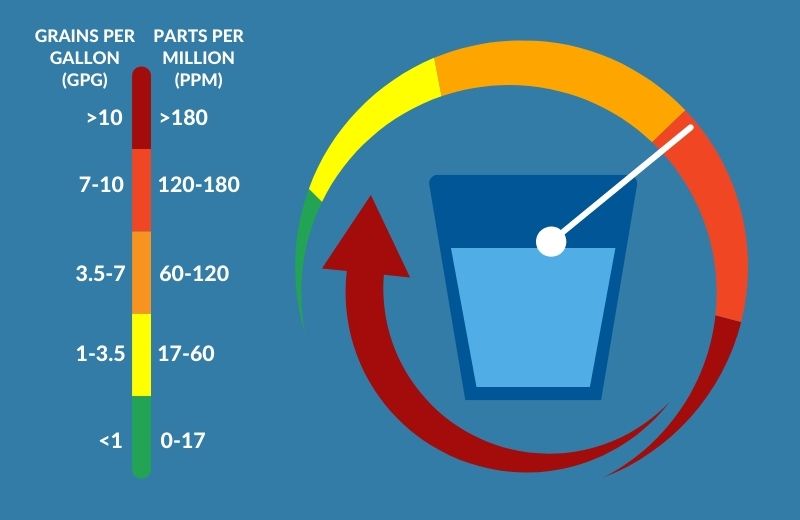
The hardness of water is measured in grains of hardness per gallon of water, or as parts per million or milligrams per liter.
- Water containing 0 to 17 parts per million of hardness minerals is considered soft.
- Water with 17 to 60 parts per million of hardness minerals is mildly hard.
- If water has 60 to 120 parts per million of hardness minerals, it’s moderately hard.
- Between 120 to 180 parts per million of hardness minerals gives water a ‘hard’ status.
- Finally, very hard water contains more than 180 parts per million of hardness minerals.
Water hardness is considered to be a problem when the concentrations of calcium carbonate and magnesium exceed 6-7 grains per gallon or 120 parts per million.
| Hardness | Grains per Gallon (GPG) | Parts per Million (PPM) & mg/L |
|---|---|---|
| Soft | <1 | 0 – 17 |
| Slightly Hard | 1 – 3.5 | 17 – 60 |
| Moderately Hard | 3.5 – 7 | 60 – 120 |
| Hard | 7 – 10 | 120 – 180 |
| Very Hard | >10 | >180 |
Benefits of Hard Water
What are some of the benefits of drinking hard water?
- Contains minerals essential for human health. We need calcium and magnesium to survive. Luckily, we also get them in our diets.
- Safe to drink and often tastes better due to alkaline mineral content. Most people prefer the familiar taste of hard, mineral-rich tap water.
- Magnesium can help prevent heart disease. It’s helpful to have it right there in your water supply.
In terms of taste and health properties, hard water is usually considered the preferred drinking water type compared to soft water. Why? Because the mineral content in hard water provides nutritional value that humans need.
As calcium and magnesium are two essential minerals to humans, their presence is a desirable benefit of drinking hard water.
Disadvantages of Hard Water
However, the disadvantages of hard water – namely its effects in your pipes and appliances – outweigh its benefits.
If your home’s water is hard, you’ll likely experience:
- Clogged pipes. Limescale clogging is especially an issue in old homes with plumbing dating back to the 1940s-1970s.
- Limescale buildup on appliances and fixtures. This may lead to reduced efficiency and potentially clog appliance pipes and drains.
- Clothes don’t get fully clean. You may notice gray marks on clothes washed in hard water.
- Water spots on dishes and faucets. Hard water leaves white spots, which are especially noticeable on glassware.
- Exacerbated eczema symptoms and dry skin. You may also experience dandruff and split ends from showering in hard water.
The dissolved minerals in hard water cause a plethora of issues. Many of these problems are caused because soap is unable to lather well due to the calcium and magnesium minerals in the water, which reduces its ability to clean clothes and surfaces.

🚰 What is Soft Water?
Soft water is water that has a very low mineral content or contains no minerals at all. Fresh rainwater is naturally soft. It is the journey the water takes once it reaches the ground that changes its structure.
📌 Note: soft water isn’t necessarily pure; it’s just mineral-free or low in minerals. Soft water may still be contaminated by other impurities affecting water quality, like chemicals and metals.
A very small percentage of households (around 15% in the US) have soft water. Anyone else who wants to achieve soft water must use a water softener system to exchange the hardness minerals with positively-charged sodium ions, which won’t cause mineral buildup.
Soft water makes tasks like washing dishes, cleaning your shower head, and washing clothes a lot easier since it doesn’t leave soap scum or mineral stains.
Benefits of Soft Water
Why is soft water so much better than hard water? We’ve shared a few reasons below.
- Increased efficiency of appliances. No more limescale clogging.
- Moisturized hair and skin. Soft water doesn’t leave soap scum, so your skin and hair are able to better retain moisture.
- Cleans clothes more thoroughly. You don’t get gray staining on your fabrics as you do with hard water.
- No residue buildup in sinks, bathtubs, or showers. Easier cleaning for you.
- Spot-free, clean dishes. You won’t need to repeatedly wash your kitchenware to achieve a shine.
When water is soft, it promotes the lathering agents in soap, improves rinsing efficiency, and has a moisturizing effect on the skin and hair.
Using soft water to wash clothes and dishes allows the water to dry without leaving mineral spots and won’t discolor your clothes. Best of all, using soft water in your plumbing won’t shorten the lives of your appliances because the surfaces will be entirely protected from scale.

Disadvantages of Soft Water
Soft water isn’t all good. While soft water has more benefits than hard water, it does have a few setbacks, too.
We’ve shared those you need to know below.
- Reduced mineral intake. Hard water only contains a small percentage of the minerals we need (most come from our diet), but drinking water that’s soft will reduce your mineral intake somewhat.
- Less appealing taste. Due to its lack of minerals, soft tap water tastes plain or even mildly acidic.
- Slippery feel. Some people find the slippery feel of salt-softened water unpleasant.
🔎 How To Tell If You Have Hard Or Soft Water
Wondering how to know for certain whether you have hard or soft water? There are a few different methods to test your water hardness.
👨🔧 One easy method is the soap test. Add warm water and a squirt of dish soap to a clear plastic bottle, then put the lid on. Shake the bottle. If the water is milky or murky, with no distinct layer of bubbles on top, it’s hard.
You can also get your water tested with a hardness testing kit, which will give you more conclusive results.

Definite Signs of Hard Water
A few definite signs of hard water are:
- Water with a slightly sticky feel
- Difficulty lathering with soap
- Film and soap scum on bathtubs
- White scale buildup around faucets and showerheads

Definite Signs of Soft Water
The definite signs of soft water include:
- Stronger water pressure
- Thick, sudsy soap bubbles when you wash your hands or dishes with soap
- A slightly salty taste (if your water has been treated with a water softening machine)
- Water with a smooth or silky feel (again, for water treated in a water softening system)
🔎 What Is The pH?
pH levels depend on the exact water hardness, but on average, hard water has a slightly above-neutral pH of 7-8, while soft water is typically 6-7, meaning it’s either neutral or slightly acidic.
Hard water contains a higher concentration of calcium carbonate, as well as other metallic minerals that make water more alkaline, such as iron, magnesium, and copper.
Soft water, on the other hand, naturally contains lower concentrations of these minerals, or it might have been treated in a water softener to remove these minerals. That means its pH levels are slightly lower because it’s missing the minerals that contribute to alkalinity.
🧰 How to Deal With Hard Water?
If you’ve discovered that your home has hard water, you probably want to fix it right away.
Water softeners are systems that turn hard water to soft water using various methods. There are several options to treat water hardness, which we’ve outlined below.
Salt-based Water Softener Systems
Considered the conventional water softener type, salt-based water softeners work by sending water through a resin bed, using the ion exchange process to replace hard mineral ions with sodium ions. This produces soft water – water that’s free from hardness minerals.
Salt-based systems are the most effective softening solution because they completely eliminate calcium and magnesium from water.

Electronic Descalers
Electronic descalers are installed on your main water pipe and create an electromagnetic field. When water passes through this field, hard water minerals are altered so that they’re no longer able to stick to surfaces. This reduces scale buildup – but it doesn’t actually remove the minerals from water.
An electronic descaler is a good option for treating moderately hard water, but we don’t recommend it for very hard water supplies.
Related: Read about the difference between descalers and water softeners
Template Assisted Crystallization (TAC)
Another salt-free water softening process is Template Assisted Crystallization, or TAC.
TAC converts hardness minerals to crystal structures that aren’t able to bind to surfaces.
Again, TAC is a process of water conditioning, not softening. However, it’s more effective than electronic descaling and the best TAC conditioners can be used to treat even very hard water.
❔ FAQ
What’s the difference between hard water and soft water?
The main difference between hard water and soft water is that hard water contains minerals, while soft water is very low in minerals or contains no minerals at all. Water that’s hard has an alkaline taste and leaves scale deposits, while soft water has a plainer taste and doesn’t cause mineral damage.
Is hard water bad for you?
No, hard water isn’t bad for you. In fact, calcium and magnesium minerals are good for your health. The biggest hard water issue is scale damage to plumbing and appliances. Showering in hard water may lead to dry skin and hair.
Is it okay to drink softened water?
Yes, it’s okay to drink water that has been softened. The lack of minerals should be fine as long as you eat a healthy diet. The exception is if you’re on a low-sodium diet and increasing your sodium levels even minimally could affect a health condition like high blood pressure.
