How often do you stop and think about what your drinking water contains? Municipal tap water is treated to meet drinking water standards, but it still contains traces of harmful contaminants that affect water quality.
This guide serves as a point of information for learning everything you need to know about drinking water contamination in the US.
Table of Contents
📝 Types of Drinking Water Contaminants
There are four types of contaminants found in the average public water system: physical, chemical, biological and radiological.
Here’s what you should know about each of them:
Physical
Physical contaminants, as the name suggests, alter the physical appearance of water. These drinking water contaminants may cause water to take on a cloudy appearance, and you may even be able to see sediment floating on the surface of the water.
They are larger than other water contaminants, and may be present in the form of sand, dust, dirt, sediment or other organic material.

Chemical
Chemical contaminants can be man-made or natural, and can affect water quality and/or pose a health risk. The most common chemical contaminant found in water is chlorine or chloramines, which is added to water during treatment to disinfect it.
Pesticides and herbicides can also make their way into water by surface run-off, and may not be adequately removed during treatment.
Biological
Any microorganism found in water is known as a biological drinking water contaminant.
Bacteria, viruses, parasites and protozoan can all survive in water after it has been treated, or may make their way into water while it travels through the pipes to your home, where they can pose a serious health risk.
Biological contaminants are particularly common in private well water sources.
Radiological
While radiological contaminants are the least commonly found in water, they still do exist in small quantities, and have the most dangerous health effects. The likes of uranium, cesium and plutonium, and radon may be present in drinking water across the country. Long-term exposure to these drinking water contaminants is considered a risk.
🚱 Types of Water Pollution
Ever wondered how drinking water supplies become polluted? According to the Natural Resources Defense Council, some of the most common types of drinking water contamination and pollution are:
Groundwater Pollution
Groundwater pollution occurs naturally. When rainwater and snow seeps through soils and rocks underground, the rain absorbs impurities from the earth, like fluoride, iron, and arsenic. Toxic chemicals and microbial contaminants may be carried into groundwater supplies in waste leached from septic systems and landfills.
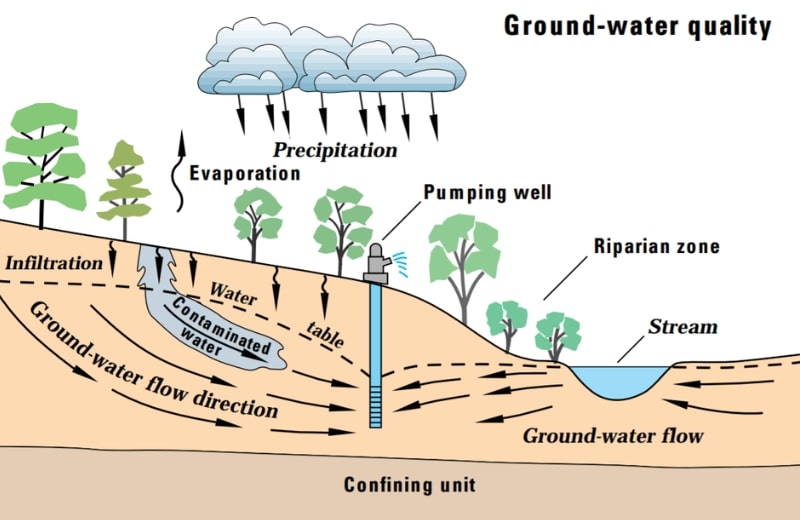
Pesticides In Groundwater by U.S. Geological Survey, 06-25-2019. Public Domain.
Surface Water Pollution
Surface water pollution occurs due to surface runoff from farms and fertilizers, dust and gas in the air, and incorrect waste disposal in industrial facilities. Toxic substances like nitrates and phosphates often contaminate drinking water as a result of surface water pollution. Blue-green algae is a source of surface water pollution that occurs naturally.
Point Source Pollution
Point source pollution is when contamination occurs from a single source. Legal or illegal wastewater discharge, chemical and oil spills, leaking septic systems, and illegal dumping are all causes of point source pollution.
Non-Point Source Pollution
Non-point source pollution is when contamination originates from multiple sources. Debris blown into waterways and stormwater or agricultural runoff are both common causes of non-point source pollution.

Transboundary Pollution
Transboundary pollution occurs when contaminated water spills into the waters in other countries. This is typically caused by a disaster, such as flooding, an earthquake, or an oil spill, or a result of w, gradual pollution from agricultural, municipal, or industrial waste.
Leaching from Water Pipes
Old drinking water systems may use pipes made from now-banned materials like lead. When these pipes corrode, they leach harmful heavy metals into the water system. The contaminated drinking water is then transported to our homes.
📌 EPA Drinking Water Contaminant Classification
In compliance with the Safe Drinking Water Act, the EPA (Environmental Protection Agency) regulates contaminants that are known to affect drinking water quality and monitors public water facilities to ensure their water is safe to drink.
Primary Contaminants
The EPA has set standards that a public water supplier must legally adhere to. These standards prevent public exposure to dangerously high levels of specific contaminants, which are known as primary contaminants.
If an impurity in water is considered unsafe, the Environmental Protection Agency has a “maximum contaminant level” for this contaminant.
A contaminant’s MCL is the highest amount it can be present in before it is considered unsafe to drink. Public water suppliers must test their water routinely and make sure they never exceed the maximum contaminant level for any of the primary contaminants, as per the EPA’s website.
The listed primary contaminants are sorted into categories: microorganisms like bacteria and viruses, disinfectants like trihalomethanes and chlorite, inorganic chemical particulates, metals like lead, organic chemical contaminants like benzene and styrene, and radionuclides like radium and uranium.
Secondary Contaminants
The EPA has also enforced National Secondary Drinking Water Regulations, or NSDWRs, for 15 contaminants. These water quality standards aren’t legal or mandatory – they’re guidelines that public water facilities can use as assistance when managing their drinking water.
The contaminants on this list are far less likely to pose health concerns, but may still have unwanted effects in high quantities.
Secondary contaminants may have cosmetic, aesthetic or technical effects on drinking water quality.
Contaminants that may affect water aesthetically are chlorine, copper, iron, manganese, aluminum copper, foaming agents, zinc, and total dissolved solids. Both fluoride and silver are secondary contaminants that may have cosmetic effects – i.e. discoloration of the teeth or skin.
Contaminants that can cause staining, scale, corrosion, and other technical effects include pH, zinc, copper, chloride, aluminum, iron, and total dissolved solids.
Contaminants of Emerging Concern
The majority of contaminants in our water have been present for decades. However, the EPA has indicated a number of emerging contaminants that are proving more and more problematic in drinking water sources in recent years.
These contaminants, known as contaminants of emerging concern, include personal care products and pharmaceuticals, microplastics and more.

🧫 Common Primary Contaminants
Microorganisms
Microbiological contaminants include:
Coliform Bacteria
Coliform bacteria, such as E. Coli and fecal coliform, are typical indicators that other harmful bacteria are present in water. While coliform bacteria aren’t a danger themselves, they’re usually a sign of contaminated water, so they have a maximum contaminant level of zero.
Cryptosporidium
Cryptosporidium also has an MLC of zero. This parasitic protozoan can cause vomiting, diarrhea, and cramps when consumed.
Giardia
Giardia, or Giardia lamblia, is a microscopic parasite that causes a stomach bug called giardiasis. It usually enters water from fecal waste and has a maximum contaminant level of zero.
Legionella
Legionella is one of the most deadly bacteria, linked to Legionnaire’s disease, a lung inflammation that can be life-threatening. Legionella occurs naturally in public water supplies and is especially susceptible to multiplying in heating systems. It has a maximum contaminant level of zero.
Viruses
Finally, viruses most commonly enter water supplies through fecal waste and can cause gastrointestinal illness, such as vomiting and diarrhea. They have a maximum contaminant level of zero.
Related: How to remove viruses from water
Inorganic Chemicals
Inorganic chemical contaminants include:
Arsenic
Arsenic, a naturally occurring chemical element, dissolves in groundwater, and is found in well water (in larger quantities) and in treated drinking water (in smaller quantities).
Certain parts of the US, like Michigan, have higher levels of arsenic in groundwater. Long-term exposure to the arsenic can cause cancer, high blood pressure, heart disease, and neurological issues.
Chromium
Chromium is a metallic element that has no taste or odor in water. It’s found naturally in soils, rocks, and animals in the environment.
There are a number of different types of chromium, and some are more harmful than others. Chromium 6 poses health risks from even low levels of exposure, while many other kinds of chromium aren’t considered harmful in trace amounts.
Copper
We need copper to stay healthy, and the compound is found in a wide range of foods, like nuts and seeds, leafy greens, dark chocolate, and some meats. But in large quantities, copper can cause a number of health issues ranging from vomiting and stomach cramps to liver and kidney problems.
Copper normally gets into drinking water that travels through old, corroded pipes, so it’s more of a problem in areas that haven’t recently replaced their water supply pipes.

Fluoride
The safety of fluoride in drinking water is still contested, with some people citing its health benefits, and others insisting that it’s harmful to health.
Fluoride is added into drinking water during treatment, at a maximum level of 0.7 mg per liter. It’s also found naturally in the ground. While it has some dental benefits (it can prevent tooth decay and cavities), a number of studies have linked fluoride contamination to illness and disease, including cancer and skeletal fluorosis.
Read More: The Best Fluoride Water Filter Reviews
Lead
Lead usually enters water through corroded plumbing pipes, especially if the water has a low pH. Many old homes have lead faucets, pipes and fixtures that can contribute to higher levels of lead in drinking water.
The maximum level of lead that should be in water is 0, as it’s a toxic metal that can cause harm to health even in small quantities. Consumption of lead in water can lead to high blood pressure, kidney damage, and even death.
Mercury
Mercury may enter water naturally from the atmosphere (such as polluted rain and snow), or from industrial and mining disposal. Some kinds of mercury also occur naturally in the ground.
The EPA’s maximum contaminant level is 0.002 (mg/L)2 for mercury. Being exposed to mercury in water above this maximum contaminant level could result in damage to the kidneys, digestive system, lungs, and immune system.
Nitrates/Nitrites
Nitrates and nitrites are particularly abundant groundwater contaminants, especially in rural areas. Overexposure to nitrates can lead to a dangerous blood disorder called methemoglobinemia. You should find no more than 10 mg of nitrates per liter of drinking water.
Disinfectants
Disinfectant contaminants include:
Chlorine
Chlorine is one of the most common contaminants affecting drinking water quality.
Chlorine is used to disinfect water and make it safe for drinking, and gives water a distinctive taste and smell that many people don’t enjoy.
Chlorine is safe to drink at up to approximately 4 mg per liter, but contamination with any more than this may result in harmful health effects or cause exacerbated allergy symptoms. Plus, chlorine is also known to produce cancer-causing disinfection byproducts.
Chloramine
An alternative disinfectant chemical is chloramine, a combination of chlorine and ammonia, which is also used in water treatment to protect against pathogens that cause waterborne illnesses.
Again, chloramine can affect water taste and smell, though it doesn’t remain in water for as long. As with chlorine, it’s safe in levels up to 4 mg per liter.
Disinfection Byproducts
Disinfection byproducts are most commonly found in chlorinated drinking water. There are around 600 known DBPs, but only a handful of these have clear data on their health effects and treatment options. Common disinfection byproducts are bromate, chlorate, chlorite, haloacetic acids, and trihalomethanes (THMs). Each of these substances has its own set of health risks.
Different disinfection byproducts have their own harmful health effects. The most common disinfection byproducts, and their health effects, are:
- Bromate: vomiting, nausea, diarrhea, abdominal pain, hearing loss, kidney effects, nervous system effects
- Chlorate: potential carcinogen, limits the blood’s ability to absorb oxygen, leading to kidney failure
- Chlorite: eye, nose, and throat irritation, coughing and shortness of breath
- Haloacetic acids: Increased risk of bladder, rectal, and colon cancer, birth defects, skin irritation, liver damage, nervous system effects
- Trihalomethanes: Increased risk of bladder and colon cancer, birth defects, reproductive problems
Organic Chemicals
Organic chemical contaminants include:
VOCs, or volatile organic compounds, are a type of chemical that can dissolve in water and vaporize in air. VOCs are incredibly toxic and can have a negative effect on the environment and human health.
Some of the most common types of VOCs are methylene chloride, benzene, formaldehyde, and tetrachloroethylene. Elevated VOCs in water can be highly dangerous as they’re known carcinogens, or cancer-forming.

Agricultural Chemicals
Agricultural chemicals, such as pesticides and herbicides, can pollute natural water sources with harmful organic material by surface runoff, and pose a health risk if high levels are consumed. When water sources are treated and sent to our homes, it’s common for trace amounts of agricultural chemical particles to linger.
Depending on the type of pesticide or herbicide, and how much of this organic material you’re exposed to, you could experience symptoms from nervous system damage to cancer, skin and eye irritation, and hormone or endocrine system disruption.
Industrial Chemicals
There are hundreds of industrial chemicals that may end up in our drinking water due to pollution of the ground or atmosphere. Organic and inorganic industrial chemicals include ammonia, oxide, nitric acid and sodium hydroxide. Petrochemicals, polymers, artificial flavors and fragrances, and industrial gases may also contaminate local drinking water supplies.
Industrial chemical contaminants are typically toxic to aquatic life in lakes & rivers. Depending on the chemical in question, consuming even low levels of these contaminants could pose a health risk, including increased cancer risk, reproductive problems, impaired eye health, and damage to the liver and kidneys.
🪨 Common Secondary Contaminants
Inorganic Chemicals
Inorganic chemical contaminants include:
Aluminum
Aluminum, a naturally occurring metal found deep in the earth’s crust, may not be noticeable in water, though contamination can cause water to take on a blue-ish tinge when it’s present in high quantities.
It’s safe to drink water that contains a very small amount of aluminum – 0.2mg/L or below – and this level of the metal can also be found in bottled water. But studies have found that exposure to too much aluminum may affect kidney and liver function.
Calcium & Magnesium
Calcium and magnesium are two of the most common minerals in water.
You’ll still have safe drinking water if it contains calcium and magnesium. The biggest problem caused by these minerals is their effect on aesthetic water quality. They’re responsible for hard water, which leaves scale deposits that are difficult to clean, damages appliances, and even affects skin and hair health.

Iron
Iron may occur in drinking water in two states: its soluble form (ferrous iron) or its insoluble form (ferric iron).
Insoluble ferric iron poses the biggest problem in contaminated water. It can stain surfaces, such as your toilet bowl, your bathtub, and even your clothes. When iron combines with bacteria, it forms a sludge-like iron bacteria that clogs up pipes and plumbing.
Manganese
If you have brownish-black staining on your appliances, don’t be so quick to blame iron as the culprit. It could be manganese, a mineral that can cause greater damage than iron in smaller amounts. Manganese may also be responsible for staining your laundry.
Sulfur
You’ll probably have a hunch if your water is contaminated with sulfur – and that’s because you’ll smell it. The sulfur odor is a little like rotten eggs, and it can make water pretty unappealing. Sulfur can also stain and damage your sinks, toilets and plumbing.
Related: Smelly water? Read our step by step guide!
💊 Common Emerging Contaminants
Organic & Inorganic Chemicals
Organic and inorganic chemicals are emerging pollutants that typically leach into lakes and rivers from refineries, factories, residential homes, and fertilizers. Contaminants include PFAS, PCPs, and pharmaceuticals.
I’ve shared more information about these drinking water contaminants below.
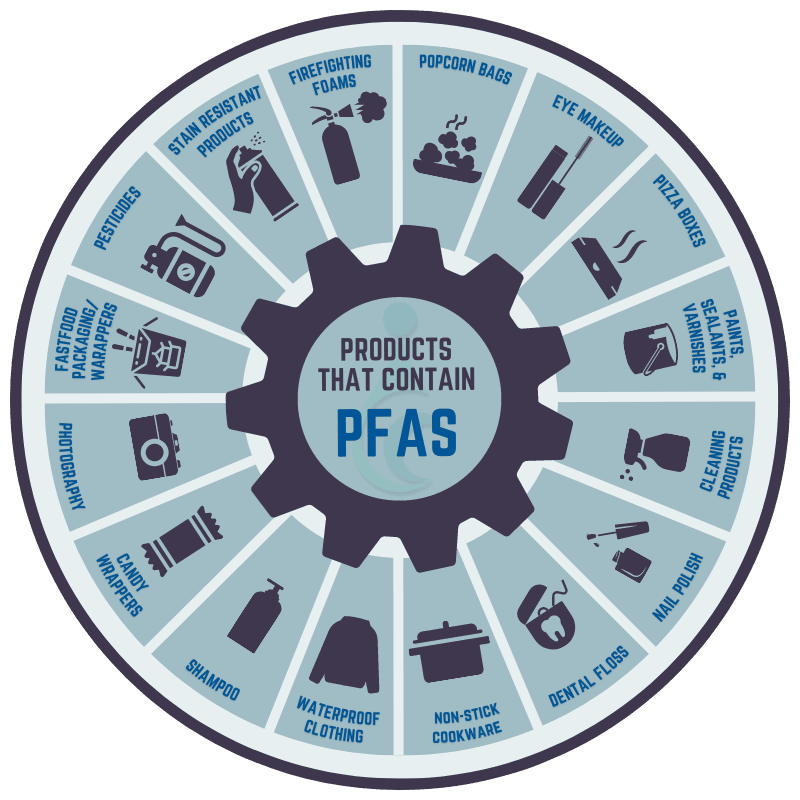
Per- and polyfluoroalkyl substances (PFAS)
PFAS are a group of manmade chemicals that were used in non-stick cooking equipment, firefighting foam, waterproof jackets, and more. They’re are known as “forever chemicals” because they linger in the atmosphere or the earth for extremely long periods of time. When consumed, PFAS may have a number of serious health effects, including reproductive and developmental, immunological, and liver and kidney effects.
Personal Care Products (PCPs)
Personal care products, or PCPs, are a group of products, including soaps, shampoo, makeup, and toothpaste, that have rapidly increased in popularity in recent years. These products, which are typically washed off skin or out of hair, pose an environmental concern, and the chemical particles they contain can affect human health.
Pharmaceuticals
Pharmaceutical pollution is another emerging water quality issue. Pharmaceutical contaminants include antacids, steroids, antibiotics, antidepressants, and anti-inflammatories. Research shows that humans are responsible for the high level of pharmaceuticals in our natural water sources.
✅ How to Remove Drinking Water Contaminants & Improve Water Quality
Keen to remove as many trace contaminants as possible from your drinking water?
Below, we’ve outlined some of the best methods for improving water quality by removing physical, chemical, biological, and radiological contaminants.
How to Treat Physical Contaminants
Some of the most common ways to remove physical particles from contaminated water include distillation, filtration (with a sediment filter, for example), and chemical disinfection. The fastest, easiest technique is whole-home filtration.
How to Treat Chemical Contaminants
Again, distillation and some types of filtration can remove chemical drinking water contaminants. Activated carbon is an effective filtration solution for removing chlorine in particular. Reverse osmosis water treatment is also very thorough at removing particulates like pesticides, herbicides, chlorine, and chloramine.

How to Treat Biological Contaminants
Biological contaminants are exceptionally small (we’re talking 0.001 microns in size), which makes them difficult to remove with the average 0.1-0.5-micron filter. Reverse osmosis membranes, which have small enough pores to trap these contaminants, can be effective for producing safe drinking water in this instance. UV filtration, which deactivates microbiological contaminants like bacteria and viruses, is another great option.
How to Treat Radiological Contaminants
Radiological contaminants are the most difficult to remove from water, and typically require a combination of filtration techniques. Ion exchange, reverse osmosis, and carbon filtration may effectively remove radiological contaminants when used together.

