Hard water causes a myriad of problems, from plumbing damage to decreased appliance efficiency.
We’ve been testing and comparing the best water softening solutions in our 10+ years of working in the water treatment industry. We’ve compiled our knowledge and data from scientific studies to produce this list of the most effective ways to soften hard water.
📌 Key Takeaways
- The two best ways to soften hard water are to use a whole-home water softener or a TAC water conditioner.
- For moderate water hardness, consider using a reverse osmosis system, an ion exchange water filter, or an electromagnetic water descaler.
- You can also boil your water to tackle temporary hardness, and use water softening products in your washing machine and dishwasher.
| Solution | Effectiveness | How it Works | Best For |
|---|---|---|---|
| Water Softener | 10/10 | Ion exchange removes calcium & magnesium | Entire home’s water supply & appliances |
| TAC Conditioner | 8/10 | Template-assisted crystallization prevents scale buildup | Entire home’s plumbing (salt-free) |
| Electronic Descaler | 6/10 | Electromagnetic pulses alter hard water minerals | Mild/moderate hard water & easy DIY install |
| Reverse Osmosis | 6.5/10 | Membrane filtration removes dissolved minerals | Moderately hard water & water purification |
| Chelating Agents | 7/10 | Chemical compounds bind to hardness minerals | Short-term water conditioning (not for pipes) |
| Ion Exchange Filter | 6.5/10 | Resin exchanges ions (point-of-use only) | Specific use point (shower, etc.) |
| Sodium Bicarbonate (Washing Machine) | 7/10 | Reacts with minerals, softens water for laundry | Washing machine only (enhanced clothes & reduced detergent) |
| Boiling Water | 6/10 | Temporary hardness minerals precipitate out | Removes scale during usage only (washing, etc.) |
| Dishwasher/Washing Machine Products | 6/10 | Additives prevent hard water effects | Appliance performance & mineral stain prevention |
Table of Contents
- 🚿 Water Softener
- 🔮 TAC (Template-Assisted Crystallization)
- 📥 Electronic Water Descaler
- ⚗️ Reverse Osmosis Membrane Separation
- 📤 Chelating or Sequestering Agents
- 🔀 Ion Exchange Water Filter
- 🪩 Add Sodium Bicarbonate to your Washing Machine
- ♨️ Boil your Water
- 🚰 Water Softening Products for Dishwashers/Washing Machines
- 📑 Final Word
- 💬 How to Soften Water FAQs
🚿 Water Softener
A water softener is the most effective solution for softening your entire home’s water supply.
Water softeners are installed at your home’s point of entry, just before your water heater. This means that your water is softened before it can enter your plumbing, so your entire water system – including all your pipes and appliances – is protected.
How it Works
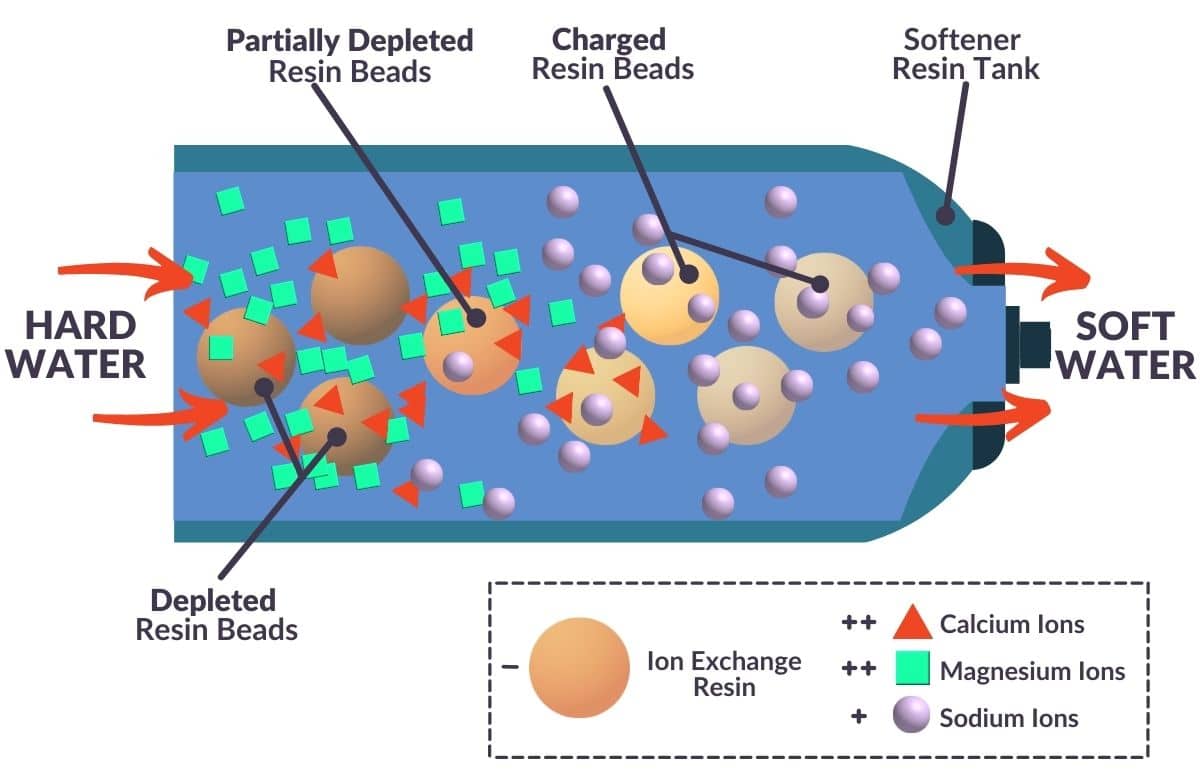
A water softener uses a process called ion exchange to remove calcium and magnesium ions from water, replacing them with sodium ions.
During this process, hard water flows through a bed of resin beads. The water hardness minerals are attracted to, and stick to, the resin. Equal concentrations of sodium chloride are released from the resin beads, balancing the water’s charge. Soft water flows out of the system.
Because hard water minerals are completely removed by an ion exchange water softener, no mineral deposits will form on your surfaces.

The Evidence
- A 2005 study on water softeners for hardness removal concluded that a hardness concentration of up to 1000 mg/L could be removed by at least 81.68% by these systems.
- Water softeners are also recommended by the EPA for homes in “areas of substantial hardness” thanks to their ability to remove the calcium and magnesium ions found in hard water.
Best For
Folks who want to eliminate hardness from their home’s water supply and protect their appliances and plumbing from limescale buildup.
👉 See our reviews of the best whole house water softener systems in 2026
🔮 TAC (Template-Assisted Crystallization)
Template-assisted crystallization water conditioners don’t technically soften water, but we’ve found that they’re the second-best solution to preventing the effects of hard water around your entire home.
TAC conditioners have the advantage of being salt-free. They also retain hardness minerals in the water rather than removing them, which means you can still enjoy the health benefits of these minerals in your drinking water.
How it Works
Water conditioners crystallize water hardness minerals with a process such as template-assisted crystallization (TAC) or nucleation-assisted crystallization (NAC). TAC conditioners contain a special media that treats water as it flows through the tank. The hardness ions adhere to the crystals, preventing them from forming scale deposits.
A water conditioner protects your plumbing system in the same way that an ion exchange water softener does, but no salt or potassium chloride is required for the process. Your water will still look, feel, and taste like hard water, but the calcium and magnesium will no longer be able to cause mineral buildup.
Although a TAC water conditioner isn’t quite as effective at eliminating scale as a water softener, it offers virtually the same performance.

The Evidence
- A study by the Water Reuse Foundation evaluated alternatives to ion exchange softeners and reported that TAC conditioning could reduce scale by more than 88%.
- A research-backed report on the University of Nebraska’s NebGuide listed TAC as an emerging technology for salt-free water softening, but noted that water “may require pretreatment” before using in a TAC system if it contains sediment or high iron and manganese concentrations.
Best For
Anyone who wants to avoid scale formation in their pipes and plumbing around their entire home without the setbacks of a salt-using softener system.
👉 Check out the best salt-free conditioners of 2026 right here.
📥 Electronic Water Descaler
Electronic water descalers, or electromagnetic descalers, are another salt-free water conditioning method.
Electronic descalers are fitted around the main water pipe leading into your home. Unlike water softeners or tank-based TAC conditioners, they don’t need to be plumbed in, which means they’re quicker and easier to DIY install.
How it Works
Electronic descalers send electromagnetic pulses into the water as it flows through the water pipe. These impulses alter the formation of hardness ions and stop them from being able to stick to surfaces as scale.
There are a couple of theories on how exactly magnetic descalers work to prevent scale formation:
- One theory is that using this conditioning technique favors the formation of aragonite (non-encrusting) instead of calcite (encrusting) mineral deposits, preventing the tough, thick limescale that folks with hard water are familiar with.
- Another is that crystals form in the water rather than on surfaces, and because of this, scale shouldn’t form (this is the same conditioning process that’s used by TAC systems).
Electronic descalers don’t waste water and don’t need salt, but they do need constant access to a power supply.

The Evidence
- A 2021 review of techniques to prevent scale concluded that electromagnetic water descalers are a “good alternative to classic water hardness reduction treatments” because they’re cheap and non-invasive, highlighting several papers that propose different theories as to how these water treatment systems work.
- The same WaterReuse study that we shared when discussing water conditioners above reported that electromagnetic water descalers reduced scale formation by about 50%.
Note: It’s important to know that electromagnetic water descalers aren’t considered a highly effective solution to hard water. For instance, this 2013 report summarized the scientific investigations into the claims of magnetic water conditioners and concluded that after “a lot of serious laboratory measurements and chemical analysis”, magnetic water conditioners were found to have no effect on water hardness.
Best For
People with a mild or moderate water hardness issue who want an easy-install, maintenance-free water conditioning solution that reduces scale but may not prevent it entirely.
👉 Read reviews of the best electronic descalers money can buy
⚗️ Reverse Osmosis Membrane Separation
Reverse osmosis is predominantly used as a water filtration solution, but RO membranes can also remove dissolved minerals, meaning that they can be used to soften moderately hard water.
Because reverse osmosis filters out most contaminants, it gives two benefits in one: soft water that’s also free from dangerous trace impurities.
How it Works
Reverse osmosis systems use a process called membrane separation to remove the majority of dissolved solids, including dissolved hardness minerals, from water.
An RO membrane typically has tiny pores (around 0.0001 microns) which allow water molecules to pass through while blocking the passage of contaminants. Water is pressurized and forced through the membrane, and the rejected impurities are flushed away as waste.
Do we personally recommend reverse osmosis as a water softening solution? Not for hard or very hard water. Hardness minerals form scale on the RO membrane, which could foul the membrane and shorten its lifespan. It’s best only to use a reverse osmosis system if you have mild or moderately hard water.
There’s also the issue that you’d need a whole-house reverse osmosis system to prevent scale formation in your entire home’s plumbing system, and most RO units are point-of-use. Whole-home reverse osmosis systems are expensive and typically cost thousands of dollars.

The Evidence
- In a 2022 study on community reverse osmosis systems for total hardness removal of groundwater, researchers concluded that RO systems produced water with the lowest mineral content (its original total hardness was 97–318 mg/L as CaCO3, which was reduced to 2.5 mg/L Ca2+, but noted that the pH of the water was “relatively low”.
- Another study conducted in 2021 evaluated the performance of existing RO drinking water stations and noted that the average hardness removal was 95.8%, which showed that RO membranes have a “higher capacity to demineralize hard water”.
Best For
Folks with moderately or mildly hard water who want to purify their water supply and are prepared for the expense and water waste that comes from using a whole home RO system.
📤 Chelating or Sequestering Agents
Chelating agents, also known as sequestering agents, are chemical compounds that address hardness in water by forming stable, water-soluble complexes with metal ions, particularly calcium and magnesium.
Chelation is another water conditioning technique that doesn’t use salt. Common chelating agents are citric acid and EDTA.
How it Works
Chelating agents contain multiple coordination sites, which surround and bond to metal ions. When they’re used for water conditioning, chelating agents bind to calcium and magnesium ions, preventing them from forming insoluble precipitates that contribute to scale buildup.
This keeps the hardness ions in the water, allowing them to be washed away and preventing them from depositing on surfaces. Chelated water typically loses its conditioning within 1-2 days, so it’s best to use this water as soon as possible.
Chelating agents are often included in commercial products like shampoos, and they’re commonly used in water softening plants.
The Evidence
- One 2021 study evaluated the use of alginate and sodium citrate (identified as a good chelating agent) for removing hardness minerals from water. The study concluded that this novel aerogel effectively removed calcium and magnesium and had maximum adsorption capacities of 62.38 and 36.23 mg/g for these minerals.
- A 2015 experimental screening of various chelating agents for calcium and magnesium removal from saline solutions concluded that EGTA and EDTA were “the most suitable” for softening Red Seawater, and EGTA was the most selective for calcium ions.
Best For
Anyone who plans to use their conditioned water within 1-2 days and is looking for a low-maintenance water conditioning method.
🔀 Ion Exchange Water Filter
Certain water filters, especially shower filters, can soften hard water. These typically use an ion exchange media to remove hardness minerals.
Just keep in mind that these filters are point of use (POU), meaning that they’re installed at the end of your plumbing and won’t protect your home’s pipes or appliances from limescale.
How it Works
Ion exchange water filters use a resin or material that can exchange ions with the water passing through it. This resin typically consists of polystyrene beads that attract positively charged ions (cations) in the water and exchange them for hydrogen ions.
The process is similar to in a water softener: as water passes through the ion exchange resin in the filter, the calcium and magnesium ions are captured by the resin, while hydrogen ions are released into the water. But once the resin is saturated, the filter needs to be replaced because it isn’t designed to be regenerated.
How effective is this method? It should work in theory, but not all water filters using ion exchange resin can remove hardness minerals. For instance, Brita filters use ion exchange resin, but they’re not tested or intended to remove hardness minerals.
If you want to be certain that an ion exchange water filter is definitely capable of softening your water, see if the manufacturer has shared test results and look for evidence of calcium and magnesium removal.
The Evidence
We couldn’t find any studies that specifically look at ion exchange water filters and their hardness reducing capabilities, but there are plenty of studies on the ion exchange process and how it works to soften water (see the studies we shared in the Water Softener section).
Best For
Folks who want to soften their water at a specific use point (like a showerhead) and aren’t bothered about preventing limescale formation in their plumbing.
🪩 Add Sodium Bicarbonate to your Washing Machine
Here’s a solution that works well for hard water issues in your washing machine.
Hardness minerals leave your clothes looking dull and feeling scratchy. If you specifically want to soften the water in your washing machine, baking soda is a good solution.
How it Works
Sodium bicarbonate, otherwise called washing soda or baking soda, reacts with hardness minerals and softens water.
When you add baking soda to your washing machine, you’ll be able to use less detergent to produce the same lather, so your washing machine doesn’t have to work as hard to keep your clothes clean.
This method should do the job, but it won’t completely eliminate hard water, and it’s only useful in your washing machine.

The Evidence
- A 2021 study on using non-industrial equipment and methods to reduce water hardness and total dissolved solids found that sodium bicarbonate could eliminate water hardness by reacting with magnesium ions. The same study noted that baking soda could also improve water taste and increase its pH.
Best For
Folks who just want to soften the water in their washing machine for enhanced laundry effects, including softer clothes and reduced detergent use.
♨️ Boil your Water
Boiling hard water removes many of the hardness minerals that cause scale. That’s why you might notice that the inside of your stovetop kettle or coffee pot has floating chunks of limescale.
This method is simple enough: just boil the water you want to soften before using.
How it Works
Boiling hard water doesn’t actually soften it in the traditional sense. Instead, it causes the temporary removal of some dissolved minerals, specifically calcium and magnesium ions, as they precipitate out of the water (a process known as thermal precipitation).
As the water is heated, calcium carbonate decreases in solubility, and solid calcium carbonates start to form. These solid particles settle at the bottom of the container, which you might be able to see as white or cloudy sediment.
Boiling your water only removes hardness minerals once you’ve taken it from a faucet. That means you can only use this water for washing your dishes, hand-washing clothes, brushing your teeth, and watering your plants.
Note: while boiling water helps to reduce temporary hardness, it doesn’t address permanent hardness, which is caused by minerals that don’t precipitate out with heat.

The Evidence
- A 2020 study examined the softening effect of boiling a soap solution, and concluded that boiling caused the volume of the soap solution to drop by almost half, indicating the removal of temporary hardness of water in the samples.
Best For
Anyone who wants an affordable method of removing temporary hardness that doesn’t require any niche equipment.
🚰 Water Softening Products for Dishwashers/Washing Machines
There are plenty of commercial products available today that can be used to produce soft water in your washing machine or dishwasher. In most cases, you just need to add a measured amount of the product to your appliance, then add your soap as usual.
In most cases, you just need to add a measured amount of the product to your appliance, then add your soap as usual.
How it Works
Water softening products designed for dishwashers and washing machines are usually sold as additives or detergents that contain specific ingredients to mitigate the effects of hard water.
These products typically contain chelating agents, sequestrants, or builders that can suspend calcium and magnesium ions in the water, preventing them from interfering with the cleaning process.
The result is an improved performance of detergents and prevention of the negative effects of hard water, such as soap scum, scale buildup, and reduced cleaning efficiency.
When you’re using a water softening product in your washing machine or dishwasher, make sure to follow the manufacturer’s instructions to get the intended effects.

The Evidence
We did some extensive research and were unable to find evidence of the effects of water softening products on preventing limescale in dishwashers and washing machines.
Our advice is to reach out to your preferred brand and ask to see data or evidence that supports their water softening claims. Manufacturers should have data on the performance of their products, and if they don’t, find another manufacturer that does.
Best For
Folks who exclusively want to prevent the effects of hard water, such as reduced appliance efficiency and mineral stains on laundry or dishes, in their dishwasher and washing machines.
📑 Final Word
If you want to mitigate the effects of hard water in your home, a whole house water softener is the best solution. But there are also methods of water conditioning, as well as ways to tackle temporary hardness or ways to protect certain faucets or appliances from hard water. The best solution for you depends on what you’re looking for.
From our own experience, we’ll say that if you want the most effective whole-house results, there are no cheap or natural water softening methods. Invest in a water softener or conditioner to enjoy softened or conditioned water throughout your plumbing and appliances.
💬 How to Soften Water FAQs
How do I get rid of hard water and iron?
One of the most effective methods of tackling iron-laced hard water is to use a well water softener. Ion exchange water softeners can typically remove around 3-4 PPM (parts per million) of iron. If your water contains more than 10 PPM of iron, you’ll need to install a whole house filter for iron removal specifically.
How do you soften water quickly?
To soften small batches of hard water, boiling your water is the quickest method, but it only addresses temporary hardness. To quickly soften the hard water in your home, the best solution is to install a water softener, which will treat the water before it travels through your plumbing.
How do you soften water cheaply?
If you can’t justify the price of a water softener or conditioner, but you still want a long-term whole-house water softening solution, the cheapest way to soften water is with an electronic water descaler. These descalers cost less than $300 and prevent hard water scale without physically removing hardness minerals from your water supply. However, they’re not as effective as salt-based water softeners.
How do I soften water without a water softener?
To soften water without a water softener, you can soften batches of your water by boiling or using water softening products. However, there are no long-term water softening solutions that address permanent hardness aside from water softener systems and salt-free water conditioners.
How do you convert hard water to soft water for a bath?
The best way to convert hard water to soft water for a bath is to install a point-of-entry water softener that will remove the hardness minerals from your home’s water supply. As a more affordable alternative, you might be able to find a bath faucet filter that contains ion exchange resin and can reduce your bathwater hardness, but most bath filters are designed to remove chlorine only.
